Clonal hematopoiesis of indeterminate potential (CHIP) describes the age-related acquisition of somatic mutations in hematopoietic stem/progenitor cells (HSPC) leading to clonal blood cell expansion and variable risk of progression to myeloid malignancy. DNMT3A is the most commonly mutated gene and while single mutations do not raise the likelihood of progression, high variant allele frequencies (VAF) and presence of co-mutations do. In myeloid diseases, the bone marrow (BM) microenvironment exhibits alterations promoting malignant transformation. However, it is unknown whether similar alterations exist in the pre-malignant niche. We set out to analyze whether human mesenchymal stromal cells (MSC) from CHIP individuals with a DNMT3A mutation ( DNMT3A mut) are altered and whether they are able to remodel the HSPC compartment in healthy hematopoiesis.
From our published cohort of previously sequenced BM samples from hematologically healthy individuals undergoing hip replacement surgery, we selected samples with either an isolated DNMT3A mut at a low VAF (2.47-6.8%, n=4, median age 73.5y, termed low-risk CHIP), or DNMT3A mut samples with a high VAF (17.5-28.5%) and at least one co-mutation (n=5, median age 73y, termed high-risk CHIP) as well as 12 age-matched samples without any mutation (n=12, median age 72y). HSPC were isolated from 3 younger healthy donors (median age 50y).
MSC were expanded by plastic adherence and subjected to multilevel characterization. Compared to age-matched healthy controls, MSC from DNMT3A mut BM samples (CHIP-MSC) exhibited higher proliferation rates, lower colony-forming-unit (CFU) fibroblast capacity, as well as reduced adipogenic and increased osteogenic differentiation potential. To understand how altered CHIP-MSC influence the hematopoietic compartment, younger healthy CD34+ HSPC were co-cultured with the different MSC subgroups for 42 hours in a low cytokine-containing medium, followed by bulk RNAseq on subsequently sorted cell populations (MSC: CD45-/CD90+; HSPC: CD45+/CD34+). We found 31 differentially expressed genes (DEG) (logFC±0.5, FDR<0.05) in CHIP-MSC compared to their age-matched counterparts. Functional analyses suggested an activation of communication towards HSPC via extracellular vesicles.
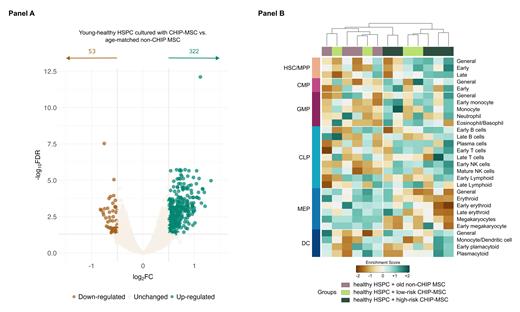
Strikingly, differential gene expression of sorted young healthy HSPC after co-culture revealed 375 DEG (322 up-regulated, 53 down-regulated; logFC±0.5, FDR<0.05) in HSPC cultured with CHIP compared to non-CHIP age-matched MSC (Figure panel A). We then used gene set variation analysis (GSVA) to analyze HSPC compartment composition after co-culture. Based on enrichment scores, unsupervised clustering identified two distinctive profiles that correlated with HSPC cultured with either non-CHIP or high-risk CHIP-MSC, while those cultured with low-risk CHIP-MSC showed an intermediate phenotype (Figure panel B). Co-culture of HSPC with high-risk CHIP-MSC resulted in enrichment of hematopoietic stem cells (HSC) and multipotent progenitors (MPP), as well as myeloid skewing represented by higher scores for common-myeloid (CMP) and granulocyte-monocyte (GMP) progenitors. Conversely, in contrast to the control group, megakaryocyte-erythroid progenitors (MEP) were underrepresented. CFU capacity was increased in healthy HSPC cultured with CHIP-MSC and multiparameter flow cytometry analysis showed a trend towards increased CMP/GMP and decreased MEP numbers, consistent with the RNAseq data.
Finally, we evaluated the BM HSPC compartment composition of a cohort of 49 CCUS patients with DNMT3A mut (VAF 1-49%, median age 76y) compared to 56 control BM samples using whole transcriptome sequencing data. Intriguingly, we observed the same profile with a myeloid shift in progenitor populations as in our healthy young HSPC exposed to high-risk CHIP-MSC.
Taken together, our results indicate that MSC from individuals harboring a DNMT3Amutation are able to instruct young healthy HSPC and influence their fate, thereby promoting a differentiation bias that shifts the hematopoietic compartment toward that associated with an aged pre-malignant state, even in the absence of DNMT3A mut HSPC. These findings suggest that focusing on the interaction between MSC and HSPC is a promising therapeutic strategy to intercept disease progression. Functional analysis of identified deregulated pathways in DNMT3Amut CHIP is ongoing.
Disclosures
Walter:MLL Munich Leukemia Laboratory: Current Employment. Benegas Coll:BioBam Bioinformatics S.L.: Current Employment. Sakuma:MLL Munich Leukemia Laboratory: Current Employment. Srivastava:BioBam Bioinformatics S.L.: Current Employment. Conesa:BioBam Bioinformatics S.L.: Current Employment, Other: Equity Ownership. Haferlach:MLL Munich Leukemia Laboratory: Current Employment, Other: Equity Ownership. Götze:Amgen: Honoraria; Servier: Honoraria; Abbvie: Honoraria; BMS: Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal